Paradis lab unearths roots of neural branching
Defects in branching may lead to epilepsy and autism
 Photo/iStock.com
Photo/iStock.comUnderstanding how neurons grow, connect and change throughout life may be the Holy Grail in treating many neurological disorders, including autism and epilepsy.
Suzanne Paradis, assistant professor of biology, and her lab recently uncovered one more clue in that quest for understanding.
The Paradis lab reported in the Journal of Neuroscience the discovery of a new signaling molecule, called Rem2, that restricts neuronal growth, or dendritic branching, in response to environmental stimuli. It is one of the first negative regulators of activity-dependent branching to be studied in vivo.
Dendrites are the primary sites where synaptic connections are formed between neurons, enabling them to communicate. Dendrites are highly branched and complex structures, and the shape of a given neuron’s dendrites is known as the dendritic arbor.
These arbors change and grow as we learn, develop memories and respond to other stimuli. The dendritic arbor needs to be highly regulated, since unchecked growth can lead to epilepsy, and too little or no growth can result in autism.
PhD candidate Amy Ghiretti led the team that studied Rem2 in live tadpoles. By exposing the tadpoles to a bright light stimulus, Ghiretti was able to chart dendritic growth in the presence and absence of Rem2.
At normal levels of Rem2, dendritic branching increased in response to light, as expected, since the tadpoles were reacting to a stimulus. However, when Rem2 levels were decreased, neurons grew out of control — at a rate up to two and a half times the normal response to light. When Rem2 was expressed beyond normal levels, the neurons showed little or no increased branching in response to the light.
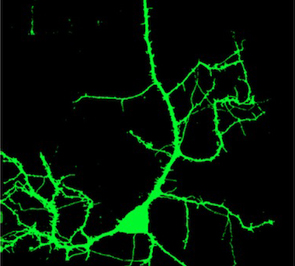 |
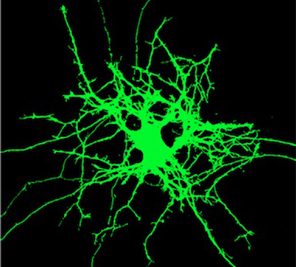 |
| Nerual branching with normal REM2 | Neural branching with REM2 decreased |
Dendrites must be able to respond to environmental changes to maintain a stable neural network, Ghiretti says. A shift in the balance of the molecules, like Rem2, that regulate the dendritic arbor — causing too many or too few branches, and thus changing the connections a neuron is able to make — may be a root cause of some neurological disorders.
“Rem2 is a new player in understanding the shape and branching of neurons,” Ghiretti says. “It’s a small piece of the puzzle but it may help us understand underlying causes of neurological disorders.”
The paper was co-authored by Brandeis University postdoctoral fellow Anna Moore, Brandeis professors Nelson Lau and Stephen Van Hooser, and collaborators Rebecca Brenner, Liang-Fu Chen, and Dr. Anne West at Duke University.
This research was funded by the Alfred P. Sloan Foundation, the Richard and Susan Smith Family Foundation and by grants from the National Institutes of Health, the Searle Scholar Foundation, the Massachusetts Life Sciences Center and the Charles H. Hood Foundation.
Categories: Research, Science and Technology





