Christine Grienberger

Assistant Professor of Biology
Research Description
Mechanisms of experience-dependent neuronal computations.
The brain’s ability to learn is critical for executing tasks that ensure an animal’s survival. My lab is interested to understand the synaptic, cellular, and circuit-level computations that allow the brain to produce these complex adaptive behaviors. To this end, we study the entorhinal-hippocampal circuit, a network of connected brain areas known to be essential for spatial learning. We use various techniques, including two-photon Ca2+ imaging, whole-cell patch-clamp recordings, and optogenetic perturbation of neuronal activity, to investigate the single-cell and population activity from hippocampal regions of mice actively engaged in a spatial memory paradigm.
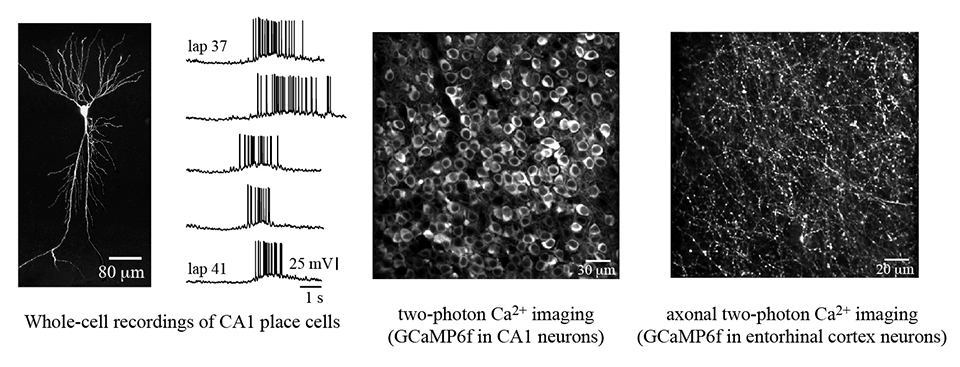
The lab pursues three main research questions:
- Individual neurons are continuously bombarded with thousands of synaptic inputs and respond by firing coherent patterns of action potentials. This input-output transformation is the basis for neuronal feature selectivity, such as the spatial tuning of hippocampal place cells. We would like to understand how learning impacts the input-output-transformations of neurons, changes their feature selectivity, and, ultimately, shapes neuronal ensemble activity. As a result, we are particularly interested in the activity of dendrites, these intricate structures where neurons receive the majority of their synaptic inputs.
- Internal representations of the external world are produced throughout the mammalian brain by the activity of many thousands of neurons, each responding to diverse environmental features. During learning, representations in the hippocampus are thought to form a cellular memory of these experiences, which can be recruited in the future to guide behavior. However, the nature of the neuronal code remains unsolved; How are representations embedded within hippocampal circuits used by the brain to produce learned behaviors? What are the cellular and circuit-level computations used to create and maintain these representations?
- Alzheimer’s disease is a disorder identified by the abnormal accumulation of amyloid-ß and tau protein in the brain. There is increasing evidence that functional circuit disruptions are among the early effects of Alzheimer’s and occur prior to the large-scale death of neurons that accompanies late-stage dementia. Moreover, the entorhinal-hippocampal circuit is among the first regions to be affected by the disease. We are interested in understanding how early-stage Alzheimer’s impacts neuronal representations in these brain areas.
Selected Publications
- Grienberger C & Magee JC. (2022) Entorhinal cortex directs learning-related changes in CA1 representations. Nature. 611(7936):554-562, DOI: 10.1038/s41586-022-05378-6.
- Magee JC & Grienberger C (2020). Synaptic plasticity forms and functions. Annual Review of Neuroscience 43, 95-117.
- Grienberger C*, Milstein AD*, Bittner KC, Romani S, Magee JC (2017) Inhibitory suppression of heterogeneously tuned excitation enhances spatial coding in CA1 place cells. Nat Neurosci. 20(3):417-26.
- Bittner KC*, Milstein AD* Grienberger C, Romani S, Magee JC (2017) Behavioral time scale synaptic plasticity underlies CA1 place fields. Science. 357(6355):1033-36.
- Grienberger C, Chen X, Konnerth A (2015) Dendritic function in vivo. Trends Neurosci. 38(1):45-54.
- Grienberger C, Chen X, Konnerth A (2014) NMDA receptor-dependent multidendrite Ca2+ spikes required for hippocampal burst firing in vivo. Neuron. 81(6):1274-81.
*Preview article in Neuron by Thomas Oertner, p1217 - Grienberger C & Konnerth A (2012) Imaging calcium in neurons. Neuron. 73(5):862-85.
- Grienberger C*, Rochefort NL*, Adelsberger H, Henning HA, Hill DN, Reichwald J, Staufenbiel M, Konnerth A. (2012) Staged decline of neuronal function in vivo in an animal model of Alzheimer’s Disease. Nature Commun. 3:774.
- Grienberger C*, Adelsberger H*, Stroh A, Milos RI, Garaschuk O, Schierloh A, Nelken I, Konnerth A (2012) Sound-evoked network calcium transients in mouse auditory cortex in vivo. J Physiol. 590(Pt 4):899-918.