Lizbeth Hedstrom
 Professor of Biology and Chemistry
Professor of Biology and Chemistry
Chair, Biology
Degrees
Brandeis University, PhD
University of Virginia, BS
Research Description
Targeted protein degradation, antimicrobial discovery; enzyme structure-function studies, mTOR inhibitors.
My laboratory uses approaches derived from both chemistry and biology. Projects include problems in inhibitor design, enzyme catalysis and protein degradation. Techniques vary with the particular project, and can entail molecular biology, organic synthesis, protein crystallography and NMR spectroscopy as well as protein purification, enzyme kinetics and mutagenesis. Our ongoing projects are outlined below. For more information, please see the group website.
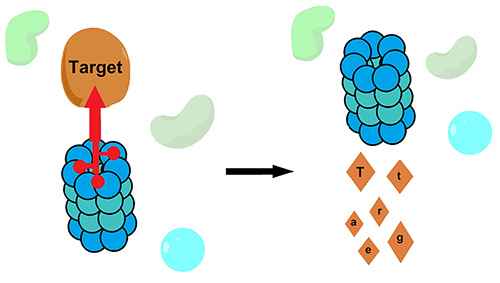 Targeted protein degradation. Targeted protein degradation is an emerging strategy in drug discovery. The rational design of degraders has generally focused on a single strategy: localization of the target protein to a ubiquitin E3 ligase. We have pioneered strategies for ubiquitin-independent degradation, where a small molecule localizes the target protein directly to the proteasome. We expect that this work will also yield important new insights into the pathways of protein quality control.
Targeted protein degradation. Targeted protein degradation is an emerging strategy in drug discovery. The rational design of degraders has generally focused on a single strategy: localization of the target protein to a ubiquitin E3 ligase. We have pioneered strategies for ubiquitin-independent degradation, where a small molecule localizes the target protein directly to the proteasome. We expect that this work will also yield important new insights into the pathways of protein quality control.
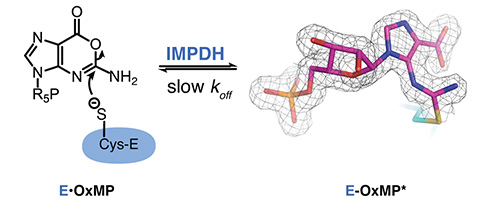 IMPDH-targeted drug discovery for infectious diseases. We exploited the unexpected divergence of the cofactor binding site in IMPDH to identify pathogen-selective inhibitors. Importantly, these compounds also display antibacterial activity. This work could lead to novel treatments for a wide variety of bacterial infections, including some of the most devastating human pathogens such as Mycobacterium tuberculosis. We are also investigating the role of IMPDH in COVID infection in collaboration with the Kadener laboratory at Brandeis. Lastly, we are investigating the mechanism of action of a potent antibacterial compound in collaboration with the Bisson laboratory.
IMPDH-targeted drug discovery for infectious diseases. We exploited the unexpected divergence of the cofactor binding site in IMPDH to identify pathogen-selective inhibitors. Importantly, these compounds also display antibacterial activity. This work could lead to novel treatments for a wide variety of bacterial infections, including some of the most devastating human pathogens such as Mycobacterium tuberculosis. We are also investigating the role of IMPDH in COVID infection in collaboration with the Kadener laboratory at Brandeis. Lastly, we are investigating the mechanism of action of a potent antibacterial compound in collaboration with the Bisson laboratory.
mTOR inhibitors. The protein kinase mTOR is the master regulator of metabolism, growth and longevity. mTOR inhibitors prolong lifespan and are used as immunosuppressive and anticancer agents. We serendipitously discovered a compound that inhibits mTOR signaling by activating the negative regulator TSC2. We are currently unraveling the mechanism of inhibition in collaboration with the Marr laboratory.
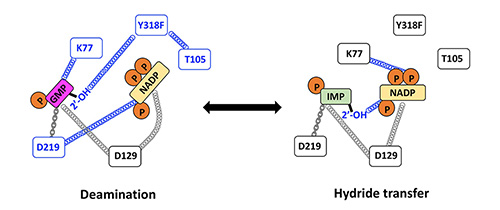 Enzyme evolution. Our current system of interest is the IMPDH/GMPR family, where we are investigating how enzymes with nearly identical active sites produce different reaction outcomes. We are also exploring how antibiotic self-resistance evolves in collaboration with the Theobald laboratory.
Enzyme evolution. Our current system of interest is the IMPDH/GMPR family, where we are investigating how enzymes with nearly identical active sites produce different reaction outcomes. We are also exploring how antibiotic self-resistance evolves in collaboration with the Theobald laboratory.
Selected Publications
- Modi, G.; Marqus, G.M.; Vippila, M.R.; Gollapalli, D.R., Kim, Y.; Manna, A.C.; Shibin C.; Maltseva, N.; Wang, X.; Cullinane, R.T.; Zhang, Y.; Kotler, J.L.M.; Kuzmic, P.; Zhang, M.; Lawson, A.P.; Joachimiak, A.; Cheung, A.; Snider, B.B.; Rothstein, D.M.; Cuny, G.D. and Hedstrom, L. The enzymatic activity of inosine 5’-monophosphate dehydrogenase may not be a vulnerable target for Staphylococcus aureus infections. ACS Infect. Dis. in press.
- Rosenberg, M.M., Yao, T., Patton, G.C., Redfield, A.G., Roberts, M.F. and Hedstrom, L. Enzyme-substrate-cofactor dynamical networks revealed by high resolution field cycling relaxometry. Biochemistry 59, 2359-2370 (2020).
- Yu, R.; Kim, Y.; Maltseva, N.; Braunstein, P.; Joachimiak, A. and Hedstrom, L. Oxanosine monophosphate is a covalent inhibitor of inosine 5’-monophosphate dehydrogenase. Chem. Res. Toxicol. 32, 456-466 (2019).
- Chacko, S., Boshoff, H.I.M., Singh, V., Ferraris, D.M., Gollapalli, D.R., Zhang, M., Lawson, A.P., Joachimiak, A., Rizzi, M., Mizrahi, V., Cuny, G.D. and Hedstrom, L. Expanding benzoxazole based inosine 5’-monophosphate dehydrogenase (IMPDH) inhibitor structure-activity as potential anti-tuberculosis agents. J. Med. Chem. 61, 4739-4756 (2018).
- Shi, Y., Long, M.J.C., Rosenberg, M.M., Li, S.; Kobjack, A., Lessans, P., Coffey, R.T. and Hedstrom, L. Boc3Arg-linked ligands induce degradation by localizing target proteins to the 20S proteasome. ACS Chemical Biology 11, 3328-3337 (2016).
- Long, M.J.C., Gollapalli, D.R. and Hedstrom, L. Inhibitor Mediated Protein Degradation. Chemistry & Biology 19, 29-37 (2012). Featured in Nature News&Views.
- Patton, G.C.; Stenmark, P.; Gollapalli, D.R.; Sevastik, R.; Kursula, P.; Flodin, S.; Schuler, H.; Swales, C.T.; Eklund, H.; Himo, F.; Nordlund, P. and Hedstrom, L. Cofactor mobility determines reaction outcome in the IMPDH/GMPR (β/α)8 barrel enzymes. Nat. Chem. Biol. 7, 950-8 (2011).
- Hansen, B.G., Sun, X.E., Genee, H.J.; Kaas, C.S.; Nielsen, J.B.; Mortensen, U.H.; Frisvad, J.C. and Hedstrom, L. Adaptive evolution of drug targets in producer and non-producer organisms. Biochemical Journal 441, 219-26 (2011).